MASS™ Medical Storage presents this document for use in understanding endoscope storage guidelines from multiple societies. The information in this guideline was compiled from current practice and published data at the time of this document’s publication. MASS™ Medical Storage assumes no responsibility for the recommendations of any society or association cited in this document.
This document is not intended to replace any guidelines or recommendations by any organization or association, but merely to provide a condensed look at current recommendations for endoscope storage.
Remember to check manufacturer’s IFUs first and foremost in order to not risk voiding the warranty on your endoscopes.
With the number of gastroenterology procedures growing every year1, proper endoscope storage is vital for patient safety.
Many societies publish detailed endoscope reprocessing guidelines, but it is equally imperative to store endoscopes safely in order to reduce the risk of contamination.
Improper storage can negate the effects of correct reprocessing by allowing for recontamination.
Given that endoscopes have such a high risk of contamination,1 medical professionals must make every effort to keep their patients’ risk as low as possible.
Cited Associations and Societies:


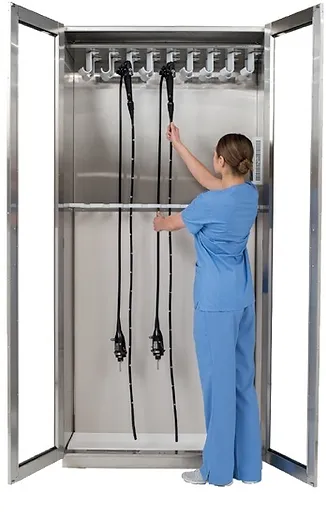
Store endoscopes in a vertical position.
Most associations recommend hanging endoscopes vertically to ensure complete drying.
EXCEPTIONS:
While SGNA recommends vertical storage, they do specify that horizontal storage is acceptable in a channel drying cabinet.1
AORN does not require this, stating that horizontal storage is acceptable if the endoscope is stored in a channel drying cabinet.2
Remember to check your endoscope manufacturer’s IFUs so as not to void your warranty.
Keep in mind: Manufacturer’s IFUs and The Joint Commission standard on scope storage do currently state that endoscopes need to be stored vertically.
Endoscopes should hang freely.
In the absence of a drying cabinet, SGNA recommends hanging endoscopes vertically and freely in order to eliminate moisture and to prevent damage to the endoscope.
Ensuring that endoscopes hang freely reduces the risk of damage from contact with cabinet walls or other endoscopes.
Removable parts should be reprocessed and stored with the endoscope.
All of these organizations recommend removing detachable components, but three are more thorough in their recommendations, stating that the endoscope’s individual pieces should be stored with it in order to allow for close tracking of use and to prevent loss of the components.
Endoscopes should be stored in a closed cabinet.
Closed cabinets help to protect the endoscopes from contamination from water droplets.
Cabinet doors also protect endoscopes from physical damage.
Storage cabinets should be equipped with HEPA filters that provide positive air pressure and circulation.
HEPA-filtered air helps prevent bacterial proliferation.
U.S. Government standards for HEPA filters currently require that aerosol penetration not exceed 0.03% (0.3 micrometer) particle size.
Do not store endoscopes in shipment cases.
Because shipment cases are not manufactured with storage in mind, they are not suitable for use as storage containers for reprocessed endoscopes.
Any carrying case that holds a contaminated endoscope should be discarded.
